Titanium in Implantology: What You Need to Know ?
Titanium as a Reconstruction and Implant Material in Dentistry: Advantages and Pitfalls
Authors: Dr. Moreno Lorso, Dr. David BrownAbstract
Commercially pure titanium (cpTi) has been a widely recognized material in dentistry due to its biocompatibility, resistance to corrosion, and impressive mechanical properties. Despite its numerous advantages, cpTi still faces some limitations as a reconstruction and implant material. This paper explores the current understanding of titanium’s material properties, its passive oxidation film, corrosion, surface activation, cell interactions, biofilm development, and associated challenges. We also discuss potential areas for improvement to enhance its clinical applications.
Keywords: biomechanics, corrosion, dentistry, implant, surface characteristics, titanium
1. Introduction
Titanium has emerged as a leading material in dentistry since the 1960s, prized for its biomechanical properties. Although there is a rising preference for metal-free restorations, failures, such as fractures and chipping, are still being reported. This suggests that metal-ceramic fixed-dental-prostheses (FDPs) remain relevant, particularly for complex oral rehabilitations where non-metallic materials may not offer sufficient strength. Titanium, especially as an implant material, continues to play a pivotal role in modern dental practice. Its introduction helped overcome the high costs of noble alloys and the potential biological risks of base metals, making it the material of choice for oral implants. However, despite its established use over five decades, research is ongoing to refine the material and optimize its properties for dental applications.
2. Material Properties of Titanium
Titanium is used either in its pure form or alloyed with other metals such as vanadium and aluminum to create lightweight, strong alloys ideal for dental implants and FDP frameworks. According to ASTM standards, cpTi is available in four grades (I-IV), each with varying levels of purity and distinct mechanical properties. Grades I and II are most commonly used in dental applications, while the higher grades find use in aerospace and medical fields.
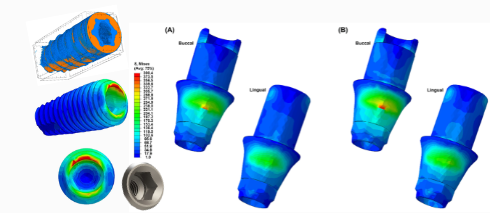
For dental implants, cpTi is prized for its biocompatibility, corrosion resistance, and strength. However, recent trends in the dental industry emphasize biomimicry, leading to efforts to make dental materials, including titanium, more compatible with the biomechanical and biological properties of bone and teeth. This approach is particularly important because the stress transfer between titanium frameworks and the surrounding dental tissues is often imbalanced due to differences in stiffness. This phenomenon, known as “stress shielding,” can lead to complications such as bone atrophy or tooth devitalization.
Research has focused on developing titanium alloys with reduced Young’s modulus to better match the stiffness of bone and teeth. For instance, β-type titanium alloys, known for their more flexible and less stiff properties, are now being explored for their potential to mitigate stress shielding. These alloys, such as Ti-6Al-4V and Ti-35Nb-4Sn, offer a promising balance between mechanical strength and flexibility, making them suitable for dental implants that more effectively integrate with bone.
3. Surface Oxide Film on Titanium
Titanium’s corrosion resistance stems from its ability to form a passive oxide film on its surface. This oxide layer, which is self-healing, protects the metal from further oxidation, making it highly corrosion-resistant and suitable for long-term use in the oral environment. The passive film’s stability is a key factor in titanium’s excellent biological performance, as it reduces the reactivity of the metal with surrounding tissues.
The oxide layer consists of hydroxyl groups that can form electric charges, depending on the pH of the surrounding environment. These charges play an important role in bonding with other materials, such as polymers, and in facilitating the immobilization of molecules. The pH of the surrounding environment influences the behavior of the titanium surface, with different types of titanium oxide exhibiting acidic or basic properties at varying pH levels. Understanding this property of titanium is crucial for improving the bonding interactions between titanium implants and dental tissues, further enhancing their clinical effectiveness.
4. Corrosion and Clinical Relevance
Materials used for dental implants are often selected for their passivity, ensuring relatively low corrosion rates compared to more reactive metals like Zinc, Magnesium, or Vanadium, which can corrode even in neutral pH conditions. In the oral cavity, while extreme acidic environments are uncommon, continuous exposure to aqueous environments, the biofilm effect, fatigue forces, and potential interactions with other metals can compromise the protective oxide layer on implant surfaces. Such conditions can result in corrosion, which can negatively impact both the mechanical integrity of the implant and the surrounding tissue. Inflammation, fretting between the implant and bone, and galvanic corrosion—particularly between titanium implants and other metallic alloys commonly used in dental procedures—can further threaten mechanical stability and clinical outcomes.
The surface properties of implants, including roughness, chemistry, and energy, directly influence tissue response by modulating protein adsorption, cell proliferation, and differentiation. Innovations in surface modification techniques have aimed at enhancing the biological performance of metallic implants. However, some surface treatments may weaken the bulk material’s mechanical properties, introducing micro-cracks and increasing corrosion rates, which can, in turn, elevate corrosion currents that impact surrounding tissues and cells.
Corrosion of metallic implants, a well-documented issue in orthopedic literature, can severely affect both the mechanical stability of the implant and the integrity of the surrounding tissue. Implant failure, such as aseptic loosening or osteolysis, may result from the release of metal ions, wear debris, or electrochemical products produced during corrosion. Studies have shown that metal ions like Ti4+, Co2+, and Al3+ can decrease DNA synthesis, mitochondrial activity, mineralization, and osteogenic gene expression in osteoblasts. Titanium particle-induced cytotoxicity has been observed in cell cultures, with higher concentrations leading to adverse effects.
While implant loosening is not as prevalent in dental literature, titanium wear particles and metal ions have been detected in tissues such as blood, liver, lungs, and lymph nodes. These particles and ions may contribute to inflammatory complications, potentially leading to macrophage activation, bone resorption, and, in rare cases, neoplasia. Titanium dioxide (TiO2), classified as a possible human carcinogen by the IARC in 2006, has been studied in animal models, though human epidemiological studies remain inconclusive. The systemic effects of corrosion remain controversial due to varying results from in vitro and in vivo studies, with some showing no significant effects of titanium ions or particles.
Electrical currents generated by corrosion are another aspect that may warrant further investigation. These currents, resulting from electron transfer during corrosion reactions, could potentially influence tissue healing, especially bone cells that are sensitive to electrical signals. Abnormal electrical signals may provide an alternative explanation for inflammatory complications and aseptic loosening in dental implants. With the increasing popularity of early implant loading, understanding the effect of these electrical signals on osseointegration and long-term outcomes is essential.
Efforts to reduce implant corrosion include developing new alloy formulations that improve both mechanical strength and corrosion resistance, as well as surface modifications designed to stabilize the oxide layer and prevent reactivity. Understanding the role of implant surface properties in the prevention of corrosion and improving clinical outcomes, particularly in cases of peri-implantitis or other implant failures, remains an area of ongoing research.
5. Titanium Surface Activation
Titanium implants, which have been used as bone anchors in dentistry for over fifty years, rely heavily on surface properties for successful osseointegration. The biological success of these implants is influenced by molecular interactions and the cellular response at the bone-implant interface. Surface modifications, including both chemical and topographical changes, enhance the rate of bone formation around implants compared to standard machined surfaces. Studies have demonstrated that ultrathin calcium phosphate coatings, for example, improve bonding between the titanium oxide and apatite nanocrystals, thereby promoting more efficient mineralization.
Surface roughness plays a critical role in cellular responses, with micrometer-scale roughness being particularly important for cellular adhesion, tissue healing, and implant stability. Various techniques, including machining, air-abrasion, acid etching, electrochemical oxidation, and laser treatment, are employed to modify titanium implant surfaces. These modifications aim to optimize the interaction between the implant surface and host tissue, particularly in the early stages of healing.
In environments with anoxic conditions, anodic oxide films on titanium may dissolve, leading to rapid corrosion. To counteract this, titanium implants are often treated with aluminum trioxide particles to enhance surface properties and improve bone integration. Coatings such as hydroxyapatite (HA) have also been explored, although challenges with adhesion and cracking during early trials have led to the development of more reliable deposition methods like physical vapor deposition (PVD) and sol-gel technologies.
Self-assembled monolayers, such as silane coatings, are being explored for their ability to modulate the hydrophilicity of titanium surfaces. These modifications aim to improve cellular adhesion and promote a faster rate of bone regeneration. Additionally, emerging strategies that involve local pharmacological release from the implant surface—such as bisphosphonates to regulate osteoclast activity—are showing promise in accelerating bone healing.
6. Titanium-Cell Interactions
The biological processes around dental implants are complex, involving a range of cellular interactions and molecular signals during inflammation, regeneration, and remodeling. In the presence of titanium implants, the implant surface serves as an osteoconductive substrate, promoting the migration of osteogenic cells and facilitating bone regeneration. Titanium surface properties—such as roughness and chemistry—dictate the sequence of cellular interactions, from protein adsorption to cellular adhesion and differentiation.
Early-stage cell adhesion to the implant surface is critical for osseointegration, with integrins playing a central role in mediating attachment. At oxidized titanium surfaces, integrin expression is upregulated, supporting faster and more effective bone healing. Studies show that the interaction between mesenchymal stem cells (MSCs) and titanium implant surfaces is crucial for the recruitment and differentiation of osteoblasts during osseointegration. Moreover, the presence of pro-inflammatory cytokines such as TNF-α and IL-1β at the bone-implant interface suggests that inflammation plays an integral role in the early stages of implant healing.
Understanding the genetic regulation of cellular behavior at the implant interface is key to improving implant outcomes. Genes related to bone formation, such as alkaline phosphatase (ALP) and osteocalcin (OC), are upregulated during the early stages of healing. Additionally, interactions between osteoblasts and osteoclasts during the bone remodelling phase are critical for maintaining bone integrity around the implant.
Research into the gene expression at the bone-implant interface is ongoing, with the goal of identifying surface properties that can accelerate or modulate healing, thus improving the long-term success of dental implants. As these studies progress, they may provide insights into how specific surface modifications can optimize the healing process and reduce the risk of implant failure.
7. Titanium-Biofilm Interactions
Titanium implants are susceptible to infection primarily due to the formation of a biofilm on the surface and compromised immune defense at the implant-tissue interface. The surface of titanium implants is biocompatible, forming a protein layer under physiological conditions that makes it conducive to bacterial colonization and biofilm formation. Biofilms are microbial communities irreversibly attached to a surface, surrounded by an extracellular matrix that protects the bacteria from the host defense system and antimicrobial agents. This makes the implant vulnerable to infection, especially in the early post-implantation phase when the local immune response is compromised. Even after tissue integration, the defense ability at the implant-tissue interface remains limited due to low blood vessel density in the area.
Research has focused on various methods to prevent bacterial contamination, such as disinfecting the implant and ensuring strict aseptic surgical protocols. Despite these efforts, bacterial contamination may still occur post-surgery or from hematogenous sources later. Studies have shown that bacteria like Staphylococcus aureus and Escherichia coli adhere more readily to titanium surfaces with nanoscale features, which promote biofilm formation. To address this, chlorhexidine coatings that adsorb onto the titanium surface and release over time have been explored. Other strategies include using alternative organic antimicrobial coatings on titanium surfaces.
Recent innovations in nanotechnology, such as nanosphere colloidal lithography and biomolecular patterning, are also being tested to improve the anti-biofilm properties of titanium surfaces.
8. Titanium Allergy
Titanium, despite its widespread use, is not universally biocompatible, and hypersensitivity to it has been increasingly recognized. Titanium implants, while generally considered inert, can release ions that may trigger allergic reactions in some individuals. Metal sensitivity, particularly in the form of a Type-IV T-cell mediated response, can cause implant failure. However, the incidence of titanium-related hypersensitivity remains underreported due to a lack of awareness and diagnostic tools. Titanium degradation products, including ions and debris, can interact with proteins and become immunogenic. Although titanium is considered relatively non-toxic, long-term clinical follow-ups are needed to assess the full impact of titanium sensitivity on implant success and failure.
9. Dental Titanium Casting
While titanium offers excellent biocompatibility and availability, it has historically been difficult to use in dental casting due to the formation of an unwanted oxide layer known as “alpha-case.” This layer impairs the bond between titanium and ceramic materials used in fixed dental prostheses (FDPs). However, advances in casting technology, including specialized refractory investments and induction melting in a vacuum, have made titanium more suitable for use in prosthetics. The alpha-case layer can now be controlled more effectively, reducing the impact on the adhesion between titanium and ceramic.
Despite these improvements, titanium’s castability remains a challenge, and further research is needed to optimize casting techniques and reduce issues such as oxidation and bonding failures. Some studies have found that alternative materials like ceramic-gold alloys may offer superior flexural strength compared to titanium-ceramic combinations, although long-term clinical data comparing these materials are still limited. The continued development of new processing methods for titanium is essential to ensure the long-term success of titanium dental restorations.
Conclusion
Titanium and its alloys have long been established as the materials of choice in dental implantology and prosthodontics, owing to their excellent biocompatibility, mechanical strength, and favorable physical and chemical properties. These materials have proven highly effective in endosseous implants, with surface modifications further enhancing their biological performance, particularly in challenging clinical scenarios. While titanium’s inherent stiffness can pose challenges, ongoing advancements in alloy composition and surface treatments have significantly improved its properties, resulting in enhanced clinical outcomes. Although difficulties in processing titanium have limited its broader use in fixed and removable prostheses, careful selection of processing methods and skilled laboratory techniques can ensure the successful application of titanium and its alloys in crowns, bridges, and other dental restorations. Despite these challenges, titanium’s role in modern dentistry continues to evolve, offering durable and reliable solutions that benefit patients globally.